Description
Características
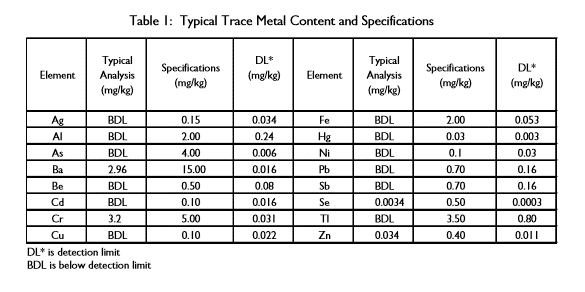
Sodium permanganate is a reagent for In Situ Chemical Oxidation (ISCO -) processes. It has been specifically manufactured for environmental applications such as soil remediation and associated groundwater. This product can be used to degrade a variety of contaminants, including chlorinated solvents, polyaromatic hydrocarbons, phenolics, organo-pesticides, and substituted aromatics. Sodium permanganate has a low concentration of metals and a pH of 5.0 to 8.0. The product is supplied with a certificate of analysis to document the pH and trace metals present.
Funcionamiento
Sodium permanganate is one of the most versatile oxidants known. Depending on the requirements of each substrate, NaMnO4 naturally oxidizes organic and inorganic compounds under acidic, alkaline or neutral pH conditions, in aqueous solutions or in the absence of water. This remarkable flexibility of the oxidant is due to its ability to act through different reaction routes and to the low enthalpy of activation that it exhibits, in the order of 21,000 to 42,000 J mol-1 (5-10 kcal mol-1), these factors that bind to the high redox potentials in any of the permanganate ion reduction states.
The specificity of the oxidations with sodium permanganate can be enhanced by maintaining strict pH control, which can be achieved, for example, by adding a buffer, such as magnesium sulphate (MgSO4).
Aplicaciones
Sodium permanganate is used for the remediation of soil and groundwater by chemical oxidation in situ or ex situ and as an active agent in reactive subsurface barriers for the treatment of: chlorinated ethenes, phenolic compounds, polyaromatic hydrocarbons, RDX, HMX and various pesticides.
FAQ
Warnings and recommendations on prevention and safety
Like any strong oxidant, liquid sodium permanganate must be handled with care. Protective equipment during handling should include face shields and / or goggles, rubber or plastic gloves, and rubber or plastic apron. If clothes look stained, wash immediately; spontaneous ignition can occur with cloth or paper. In cases of significant exposure, use the appropriate NIOSH-MSHA dust or mist respirator. For more details do not hesitate to consult our SDS.
Storage
Avoid contact with acids, peroxides, and all combustible or easily oxidizable organic materials, including inorganic oxidizable materials and metal powders. With hydrochloric acid, chlorine gas is released. Liquid sodium permanganate is not combustible but will support combustion. It can decompose if exposed to intense heat. Fires can be controlled and extinguished by using large amounts of water. For more details do not hesitate to consult our SDS.
Delivery format
- 20 L tanks of concentrated sodium permanganate solution
- 208 L drum of concentrated sodium permanganate solution on pallet, strapping and shrinking.